Carbon element valence electrons1/17/2024  
In this way carbon is a very important element of the human body. When we breathe in oxygen gas (O 2), it combines with the carbon (C) of the body and finally carbon dioxide (CO 2) is formed which comes out of the body when we exhale. The molecules of proteins, lipids, carbohydrates, nucleic acids, contain carbon in it. 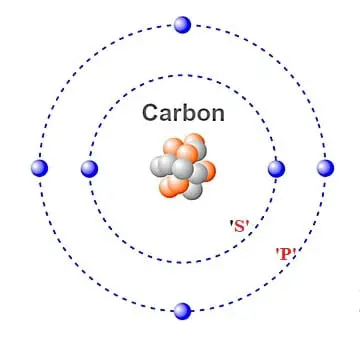
It is present in our body cells in the form of sugars like glucose.Ĭarbon enters our body from the food we intake.Ĭarbon is present in high quantities in foods which are rich in carbohydrates. In other words, carbon consists of around 18.5 % of mass in the human body.Ĭarbon is the basic building block of our body. 
In the human body, around 18.5 % of mass is because of carbon. Yes carbon is present everywhere, in all living organisms. Note: Not all the diamonds are colorless. Also the structure of diamond is such that most of the light passes through it. And so, these bonded electrons do not absorb any radiation. While in diamond, the adjacent atoms are covalently bonded with each other. That means graphite has delocalized electrons which are responsible for electric current.Īlso, these delocalized electrons absorb most of the light.Īnd as the light is absorbed, it appears shiny black in color. You might be knowing that graphite is a good conductor of electricity. Now, for graphite and diamond, the entire concept is based on how the electrons are bonded with each other. If the object absorbs all the light, then it will appear completely black.Īnd if the object does not absorb any light (or it reflects all the light), then it will appear completely white. The color of any substance depends upon the light absorption by the object. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
